The VIDAS® Offer
Trusted around the world, VIDAS® has been recognized for over 3 decades as a leader in on-demand immunoassay testing, with one of the broadest installed bases of benchtop immunoanalyzers. The success of VIDAS® is based on the quality of its reagents and the robustness and ease of use of its instruments, associating the single-test concept with ELFA technology.
The comprehensive VIDAS® menu offers quality and confidence in results with more than 80 available tests. Reliable results allow you to make the right treatment decisions for a wide range of acute medical conditions and infectious diseases including, but not limited to cancers, infertility, pregnancy, and thyroid disease.
Built to withstand the test of time, VIDAS® provides enduring peace of mind and stands as a hallmark of unparalleled quality. With constant research into new parameters offering high medical added value, innovation is the cornerstone of VIDAS®. We are constantly innovating in an effort to offer instruments with more simplicity and automation resulting in enhanced lab productivity for you.
Disclaimer: Product availability varies by country. Please consult your local bioMérieux representative for product availability in your country
-
VIDAS® KUBE™
Stackable benchtop automated immunoassay solution
VIDAS® KUBE™ is designed to preserve everything labs appreciate about the VIDAS® Solution combined with advanced technology. VIDAS® KUBE™ is a truly flexible, cost-effective automated immunoassay solution providing fast results in complete confidence.
-
VIDAS® 3
Trusted. Robust. Innovative.
With its advanced immunoassay technology and optimized testing protocols, the fully automated VIDAS® 3 benchtop immunoanalyzer empowers your lab with confidence, offering the precise results you need, when you need them.
-
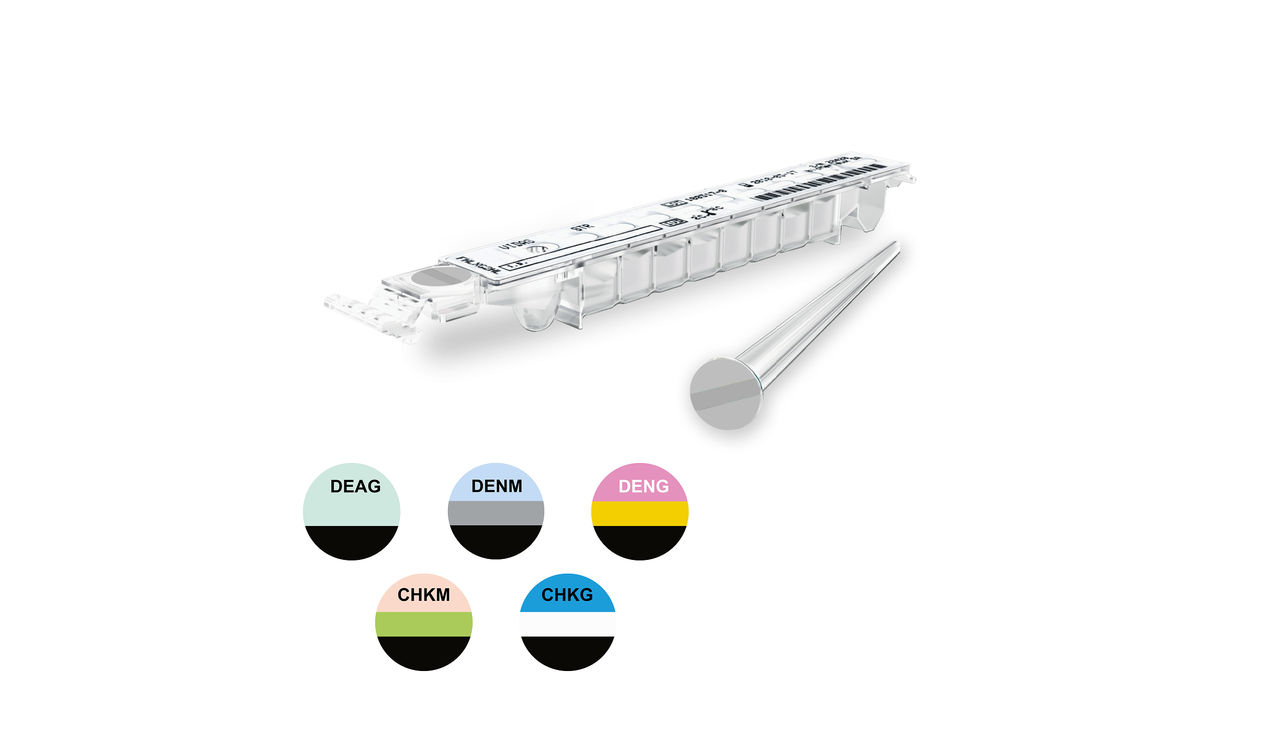
VIDAS® Arboviruses Panel
Suspecting an Arbovirus? Think Dengue and Chikungunya
Our VIDAS® offer of immunoassays directed against arboviruses diseases improve access to medically important automated diagnostic tests.
-
VIDAS® B•R•A•H•M•S PCT™
Important Biomarker for Improved Patient Management
Detects procalcitonin, a biomarker that aids in the risk assessment for progression to severe sepsis and septic shock. PCT also aids in decision making on antibiotic therapy for patients with lower respiratory tract infections (LRTI).
-
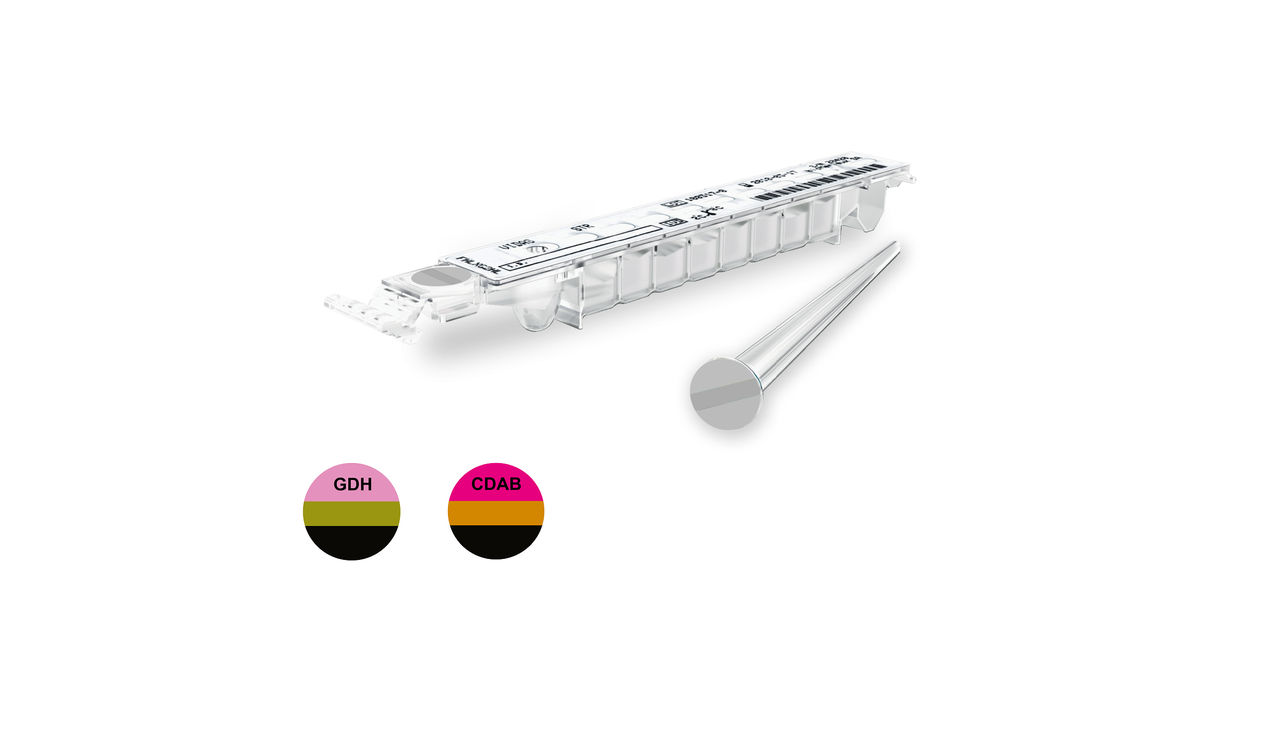
VIDAS® C. difficile Panel
Rapid identification of C. difficile infection
The VIDAS® C. difficile GDH and TOXIN A & B (CDAB) are two complementary tests used as an aid in the diagnosis of C. difficile associated disease. NEW IVDR: VIDAS® GDH and VIDAS® CDAB are already CE Marked under the new In Vitro Diagnostic Medical Devices Regulation (EU) 2017/746 (IVDR).
-
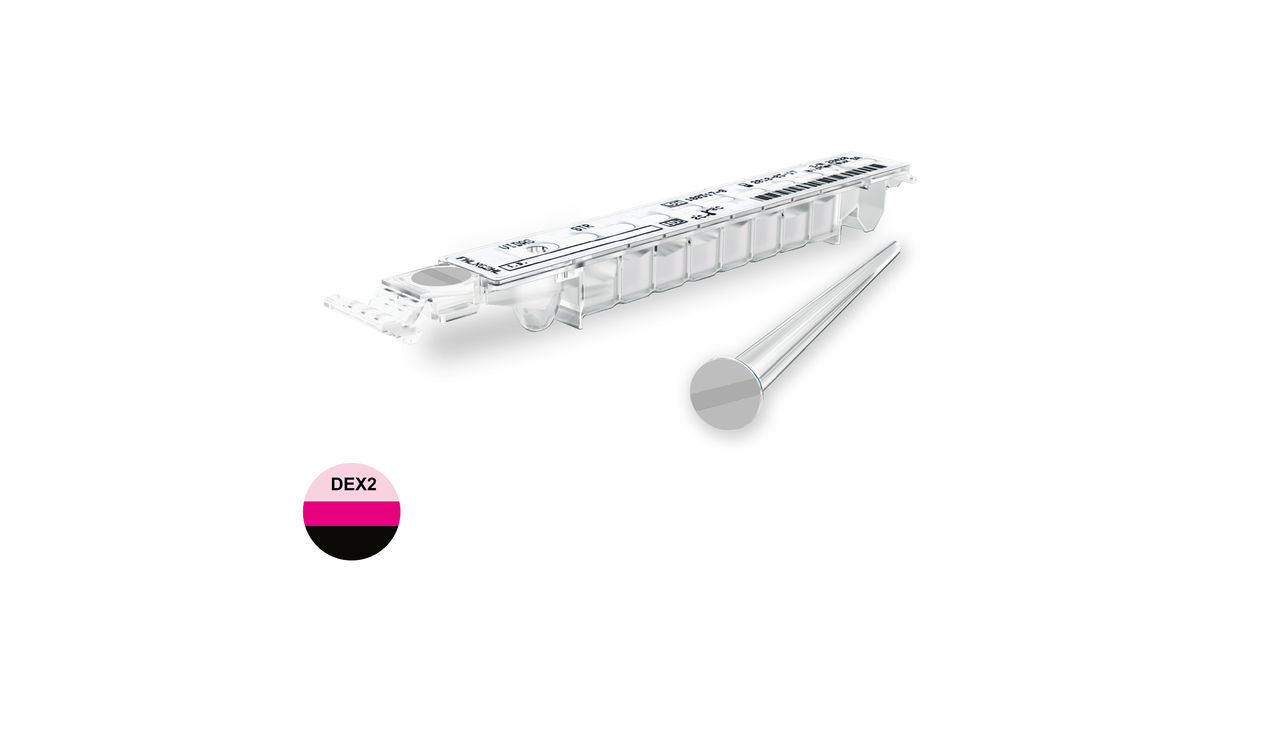
VIDAS® D-DIMER EXCLUSION™ II
Safe Exclusion of Venous Thromboembolism
VIDAS® D-Dimer Exclusion™ II is a highly-sensitive automated D-Dimer assay. Used in conjunction with assessment of clinical pretest probability, it allows safe exclusion of deep vein thrombosis (DVT) and pulmonary embolism (PE) in both low and intermediate suspected outpatient risk groups in just 20 minutes.
-
VIDAS® High sensitive Troponin I
Optimize the Management of Acute Coronary Syndrome (ACS)
The VIDAS® High sensitive Troponin I assay is intended to be used as an aid in the diagnosis of myocardial infarction delivering a diagnosis in only two hours and for the risk stratification of patients with acute coronary syndrome.
-
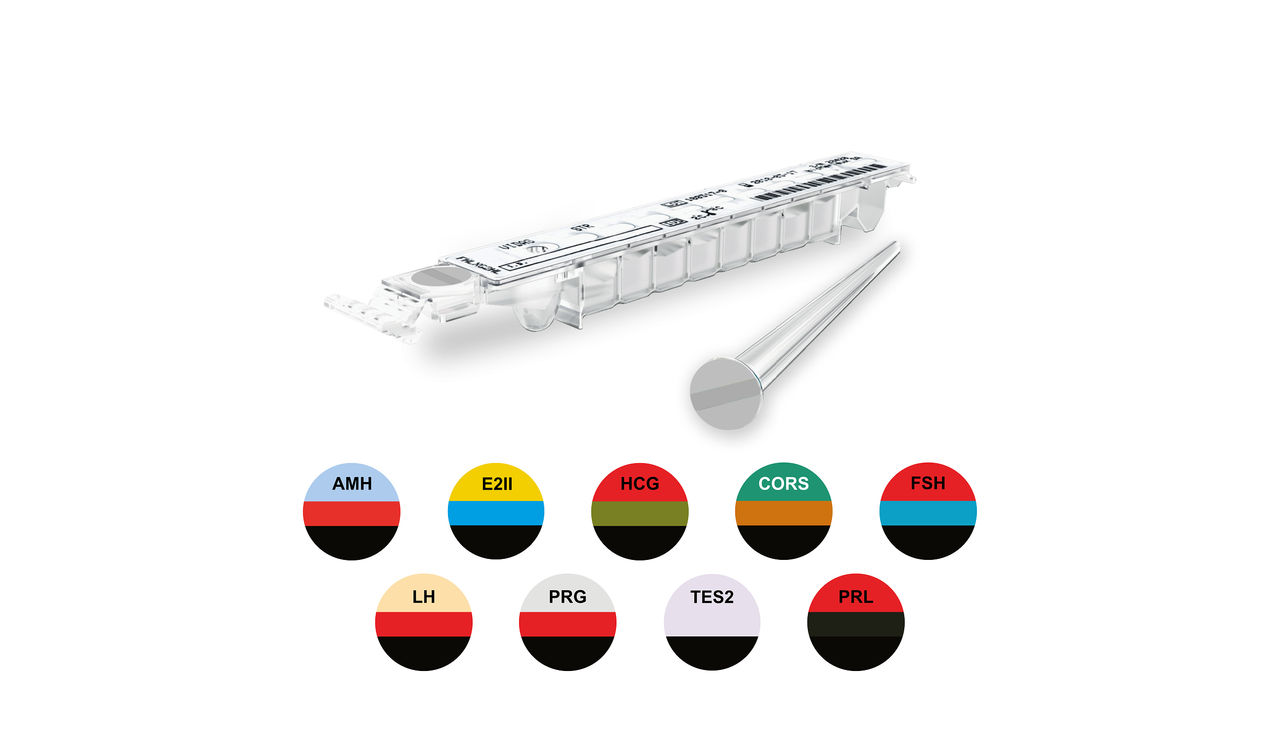
VIDAS® Hormone & Fertility Assays
A Full Panel for the Diagnosis and Monitoring of Infertility and Adrenocortical Disorders
The VIDAS® Fertility panel provides key diagnostic tools for the investigation of reproductive hormone dysfunctions such as precocious and delayed puberty, amenorrhea, hirsutism, hyperprolactinemia, perimenopause and menopause, hypogonadism, gynecomastia, and azoospermia
-
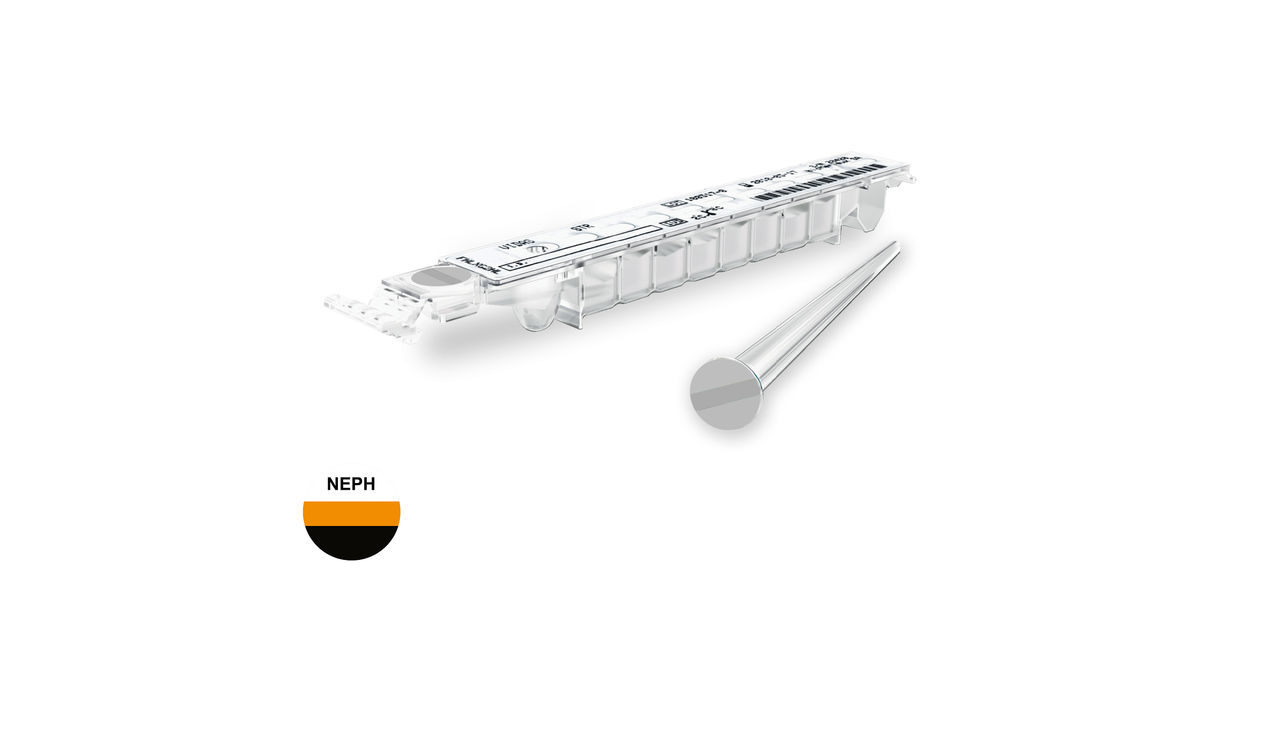
VIDAS® NEPHROCHECK®
Reveal Kidney Stress Early. Drive Better Outcomes.
VIDAS® NEPHROCHECK® measures urinary TIMP-2 & IGFBP-7, specific urinary biomarkers for early risk assessment of moderate to severe Acute Kidney Injury (AKI). The VIDAS® NEPHROCHECK® assay is intended to be used in conjunction with clinical evaluation as an aid in the risk assessment for moderate or severe acute kidney injury (AKI) in acutely ill patients.
-
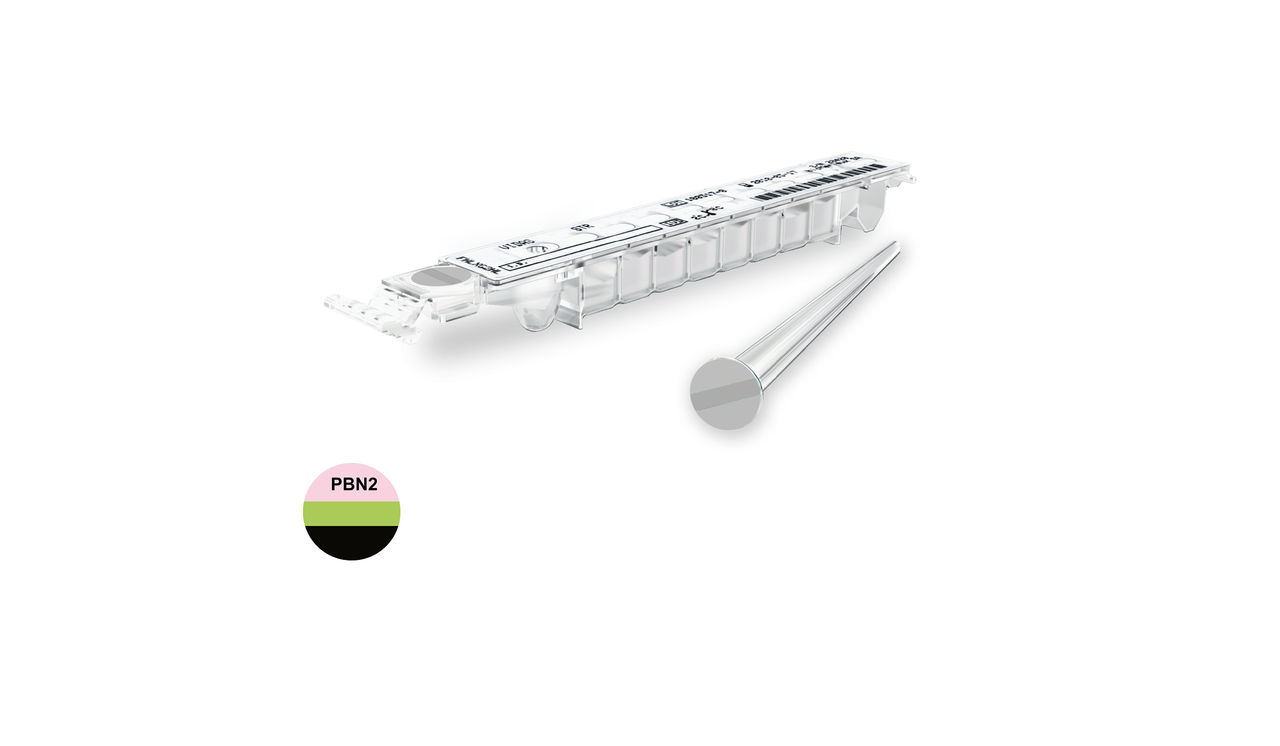
VIDAS® NT-proBNP 2
Exclusion or Confirmation of Acute Heart Failure
VIDAS® NT-proBNP2 is a useful tool for rapid and clinical assessment and diagnosis of patients with symptoms of suspected heart failure.
High sensitivity and specificity allows initiation of timely, appropriate treatment.
Cost-effective tool adapted to emergency situations.
-
VIDAS® SARS-COV-2
Two immunoassay tests to detect IgM & IgG antibodies
VIDAS® SARS-COV-2 IgM and VIDAS® SARS-COV-2 IgG II automated qualitative assays rapidly detect antibodies to help identify individuals with previous exposure to SARS-CoV-2.
-
VIDAS® TBI (GFAP, UCH-L1)
RULES-OUT Intracranial Lesions for mild Traumatic Brain Injury
Fast all-in-one test kit based on blood brain biomarkers to predict absence of intracranial lesions in mild Traumatic Brain Injury patients and objectively assess the need for a head CT-scan.
-
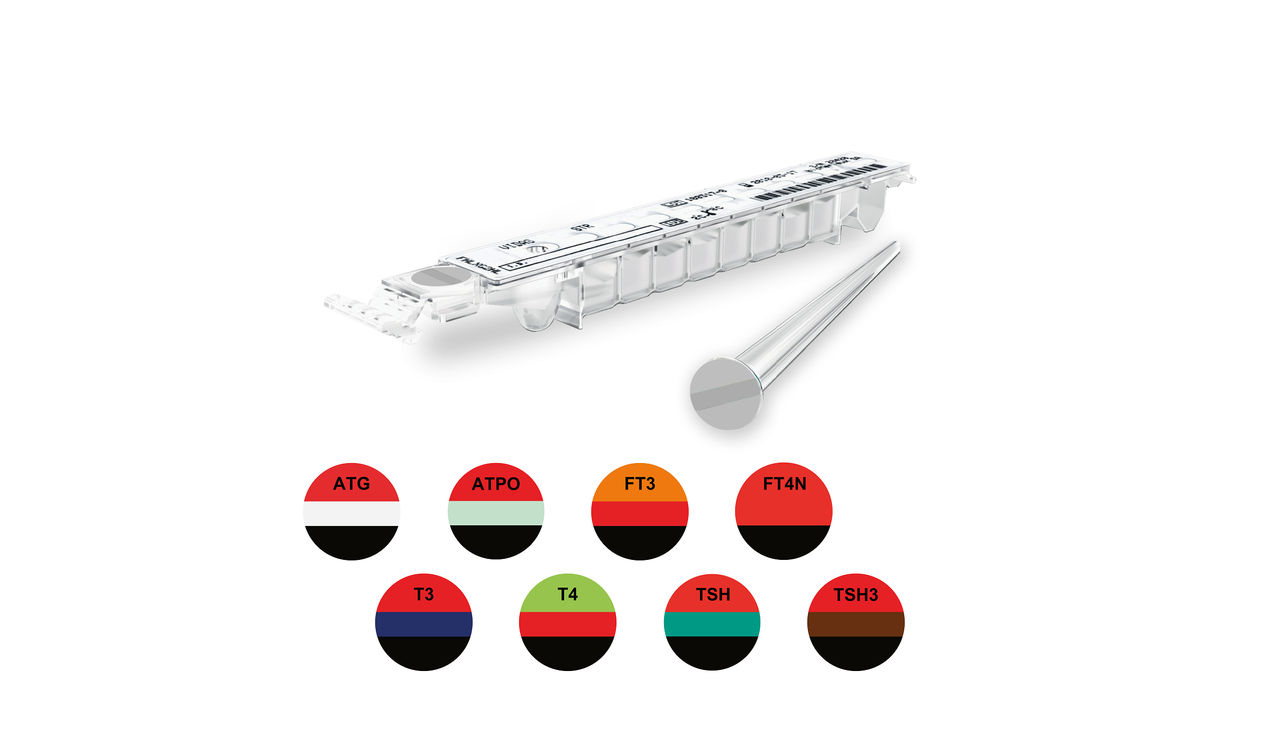
VIDAS® Thyroid Assays
A Full Panel for the Investigation of Thyroid Diseases
The VIDAS® Thyroid panel includes 8 automated tests to aid clinicians in the diagnosis and treatment monitoring of thyroid disorders.
-
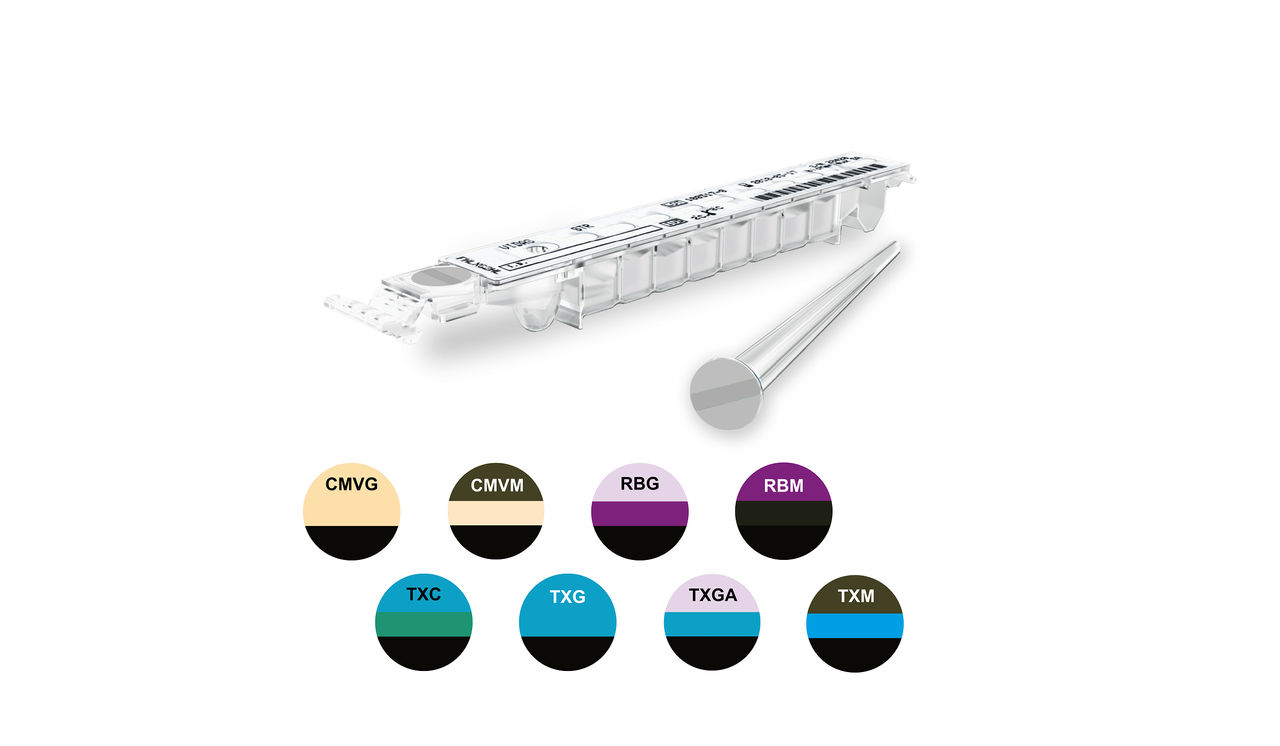
VIDAS® ToRC Panel
Serenity throughout Pregnancy
The VIDAS® ToRC panel included 9 automated tests for the screening and diagnosis of toxoplasmosis, rubella, and cytomegalovirus (CMV) infections.
-
MYACUTECASE™ Mobile App
All the information you need for confident clinical decision- making with the VIDAS® Emergency & Critical Care assays
Designed for healthcare professionals, MYACUTECASE™ is a first-of-its-kind app. Used in conjunction with clinical assessment and other laboratory findings, it provides comprehensive biomarker information and useful tools for:
Useful Resources on VIDAS® SOLUTIONS
bioMérieux
VIDAS® Journey